Japan approves stem-cell treatment for Parkinson
Japan’s health ministry is fast-tracking iPS cell therapies under conditional licenses that move commercial medicine to market on smaller-than-usual trial evidence.
Mar 6, 2026
Sources
Summary
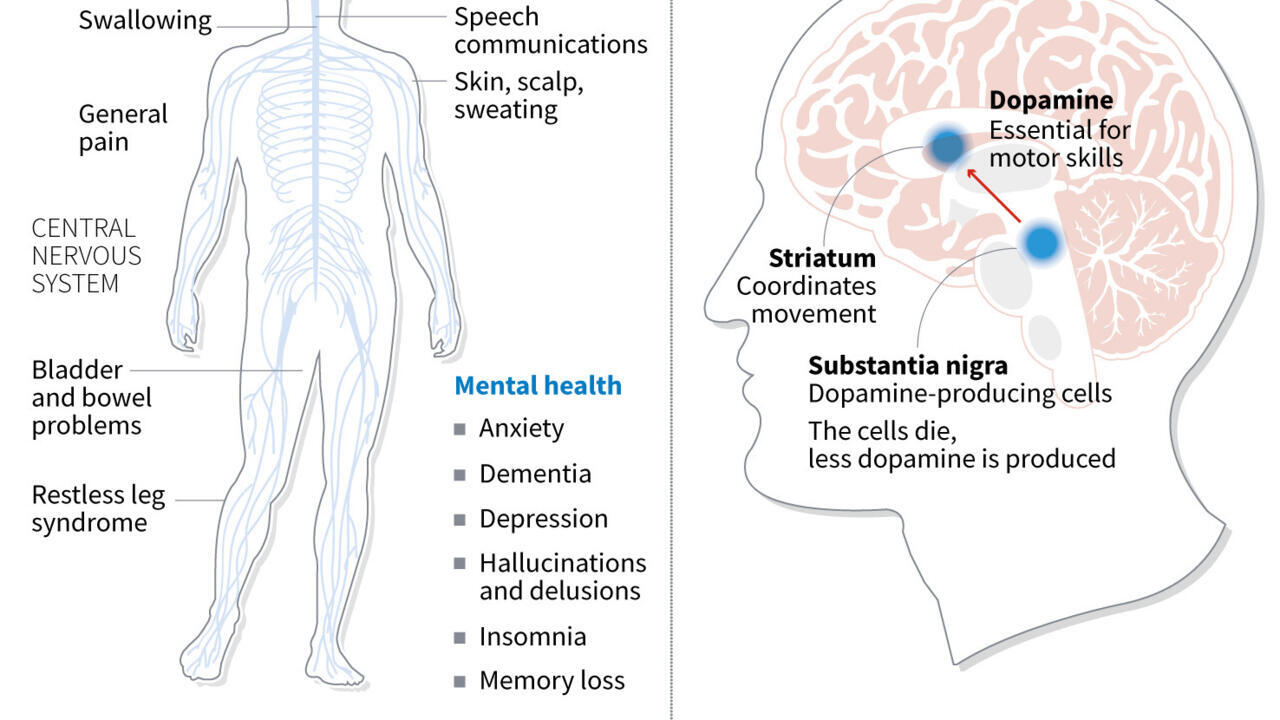
Japan’s health ministry approved conditional, time-limited manufacture and sale of iPS cell-based therapies for Parkinson’s disease and heart repair. The decision uses a fast-track approval system that relies on efficacy and safety assessments drawn from fewer patients than ordinary clinical trials. The approvals could place the first commercially available iPS cell products on the market as early as this summer, accelerating patient access while locking in a precedent for provisional licensing in regenerative medicine.
Detail
<p>Sumitomo Pharma announced it received approval to manufacture and sell Amchepry, a Parkinson’s disease treatment that transplants stem cells into a patient’s brain. Japan’s health ministry also approved ReHeart, heart muscle sheets developed by startup Cuorips, which media reports said can help form new blood vessels and restore heart function.</p><p>Reports citing the health ministry said both products could reach the market and patients as early as this summer, described as the world’s first commercially available medical products using induced pluripotent stem (iPS) cells.</p><p>Sumitomo Pharma said Amchepry received “conditional and time-limited approval” under a system designed to speed patient access. The Asahi newspaper described this as a provisional license issued after safety and efficacy were judged using data from fewer patients than typical drug trials.</p><p>A Kyoto University-led study followed seven Parkinson’s patients for two years after implantation of five million or 10 million iPS-derived dopamine-cell precursors on both sides of the brain, reporting no major adverse effects and symptom improvements in four patients.</p>